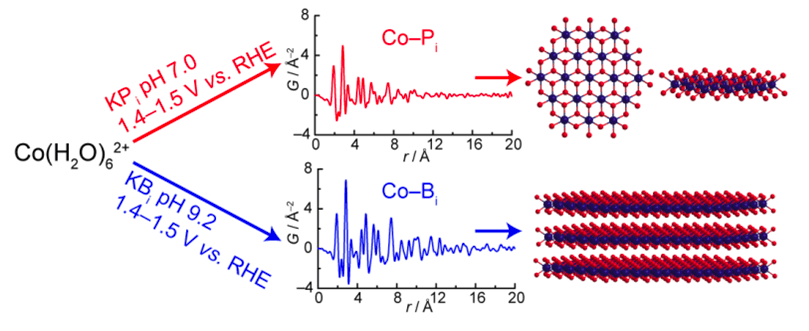
It will take state-of-art nanoparticle catalysts to harness sunlight to split water, a holy grail of energy research, into hydrogen and oxygen, green fuels with the potential to satisfy the ever-increasing demand for energy. Due to the catalysts’ nanostructured nature, their structures are unknown, which poses a great challenge for understanding their function. Recently, Billinge group post-doc Christopher Farrow, now working at Enthought Scientific Computing Solutions, utilized x-ray PDF to provide the first ever structure function correlation results in cobalt thin-film catalysts. The collaboration, with the group of Prof. D. Nocera at Harvard University, interrogated the intermediate structures of two cobalt oxide catalysts that were electrodeposited in borate and phosphate solution, respectively. The films from the borated solution were more ordered than the phosphate derived films, and with increasing film thickness, borate films were more catalytically active than the phosphate counterpart. The atomic-scale structure of the two types of nanoparticles was the same, but the nanoscale and mesoscale structures were different, and critical to the performance, as demonstrated by this work. The manuscript was published in the April 2, 2013, online edition of the Journal of the American Chemical Society (http://pubs.acs.org/doi/abs/10.1021/ja401276f) and was recently highlighted by NSLS on the Brookhaven website (http://www.bnl.gov/newsroom/news.php?a=24444).
-
Subscribe
Subscribed
Already have a WordPress.com account? Log in now.